GH3
Cat.No.: CSC-C9384L
Species: Rattus norvegicus (Rat)
Source: Pituitary Gland
Morphology: Epithelial
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
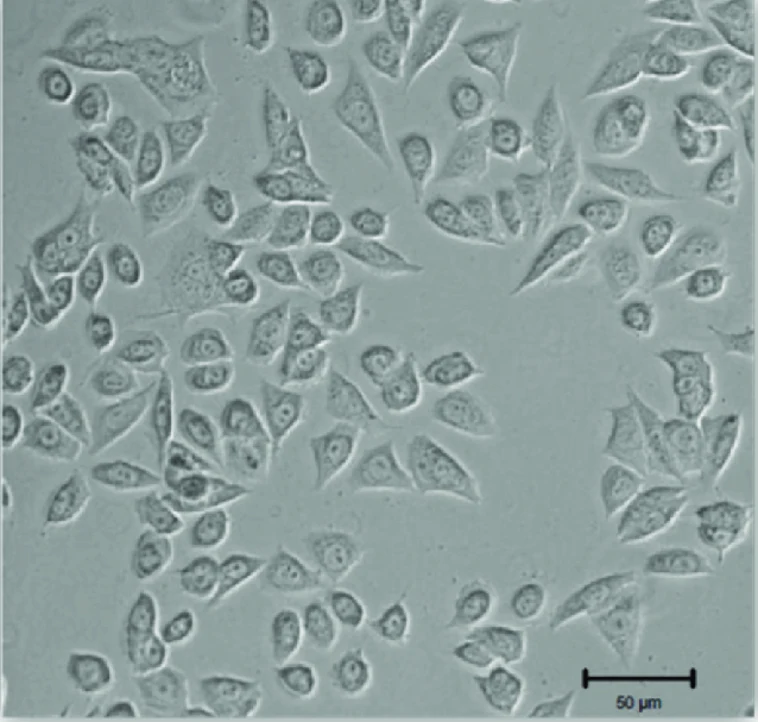
GH3 cells are rat pituitary adenoma cell line which was originally isolated from a functional pituitary tumor. This cell line has been extensively characterized for synthesis and secretion of pituitary hormones, particularly prolactin (PRL) and growth hormone (GH). GH3 cells have been used as a classic pituitary tumor cell line for numerous in vitro studies related to endocrine and neuroendocrine function.
GH3 cells are adherent, polygonal to rounded epithelial-like cells that demonstrate stable growth rates under normal culture conditions. These cells express classic pituitary-specific transcription factors as well as hormone-processing machinery and maintain differentiated functions after being propagated in vitro. Like native pituitary cells, hormone release from GH3 cells is regulated by physiological stimuli such as thyroid hormone or pharmacological stimulation with dopamine agonists or growth factor receptors.
Molecular studies using GH3 cells have been used to understand regulation of pituitary hormone gene expression, signaling pathways that regulate endocrine secretion and proliferation, and pituitary tumor growth. These cells are also routinely used as an in vitro model for testing endocrine-disrupting chemicals as well as identifying pituitary-mediated toxicological endpoints, specifically relating to thyroid hormone and dopamine signaling.
Thyroid Hormone Disrupting Potentials of Benzisothiazolinone in Embryo-Larval Zebrafish and Rat Pituitary GH3 Cell Line
Benzisothiazolinone (BIT), One of the most widely used antimicrobial agents in consumer products, has frequently been detected in the water environment. The present study was conducted to determine the adverse effects of BIT on the thyroid neuroendocrine system of zebrafish embryos/larvae. Significant coagulation and hatching delay were observed in embryos exposed to 30 μg/L of BIT, which in turn remarkably decreased hatchability and larval survival.
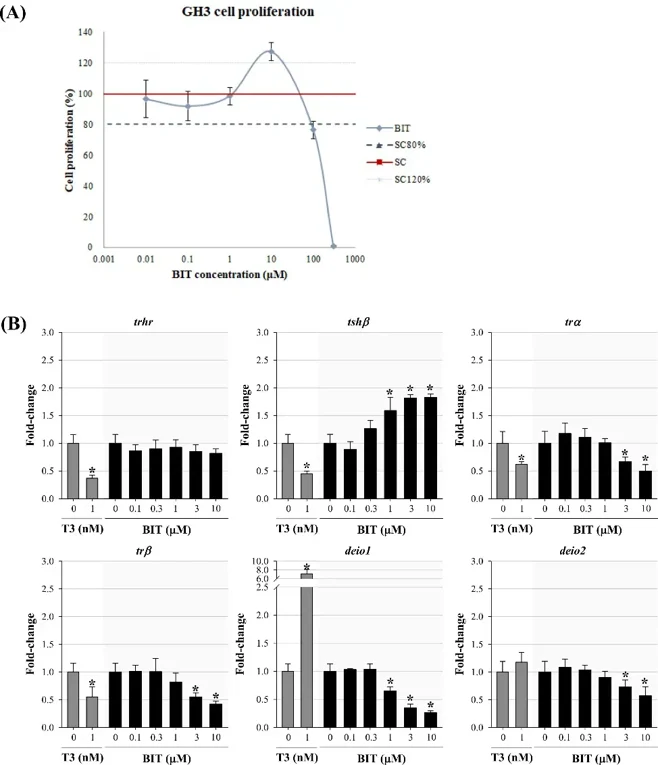
Then, rat pituitary (GH3) cell line was employed to support the underlying mechanism of thyroid hormone disrupting effects. Fig. 1A shows the effects of BIT by 48 h exposure in the cell proliferation assay. BIT stimulated cell proliferation to the greatest extent at 10 μM. Since rapid cell death was observed at ≥ 100 μM BIT through cell proliferation assay, the cells were exposed at concentrations of 10 μM or less for gene analysis. Significant up-regulation of tshβ and down-regulation of trα, trβ, deio1, and deio2 genes were observed following exposure to BIT (Fig. 1B). Their observations suggest that BIT can decrease the level of thyroid hormones by influencing central regulation, receptor binding, and deiodination.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells