Rat Embryonic Hematopoietic Stem Cells
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Rat Embryonic Hematopoietic Stem Cells (HEpSCs) are hematopoietic stem cells that are isolated from rat embryos at early developmental stages. HEpSCs are an early population of hematopoietic stem cells which give rise to the hematopoietic system during embryonic development. These cells have robust self-renewal capacity and multilineage differentiation potential and are able to generate all mature blood cell lineages including red blood cells, white blood cells, and platelets. In the embryo, hematopoietic stem cells are derived from hemogenic endothelium in the aorta-gonad-mesonephros (AGM) region, fetal liver, and yolk sac and expand and mature as they colonize the bone marrow.
HEpSCs express hematopoietic stem and progenitor cell markers and are able to form colonies in hematopoietic assays. These cells can also differentiate into multiple hematopoietic lineages in vitro when given the correct culture conditions along with appropriate cytokines and growth factors. Due to their stage of derivation, they have been used to study early hematopoiesis, stem cell specification, and commitment to a particular lineage. HEpSCs are commonly used in biomedical research to study molecular processes involved in hematopoiesis and regulation of stem cells. They can be used to study embryonic hematopoiesis, gene regulation during embryonic development, and hematopoietic stem cell differentiation.
The Pre-Implant Model is Highly Responsive and Sensitive to the Embryotoxic Effect Induced by RA, TBBPA and PFOA
International guidelines for developmental toxicology studies require ≥60 pregnant rats per molecule, while current in vitro methods show poor translational power. Quadalti et al. proposed an innovative rat embryonic stem cell (RESC)-based platform for wide-scale screening, mimicking pre-implant (undifferentiated pluripotent cells) and post-implant (neuroectodermal differentiation) stages.
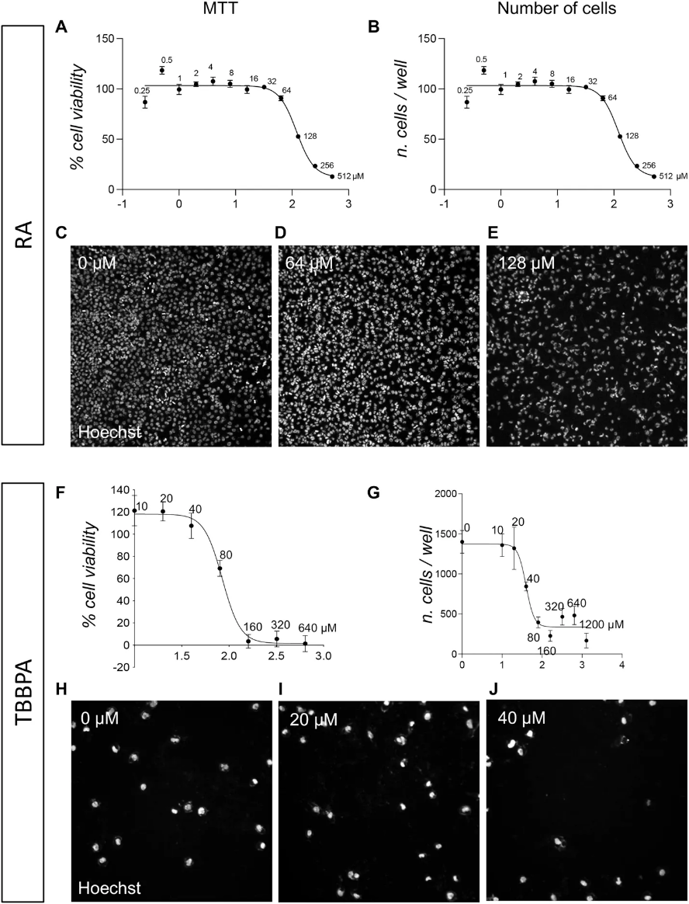
Three compounds (RA, TBBPA, PFOA) were tested in the pre-implant model. Acute toxicity (24 h) was assessed by MTT assay and high-content screening (HCS)-based cell counting. MTT viability assay yielded an RA IC₅₀ of 119.9 µM (95% CI 101.2-149.3 µM; Fig. 1A). Although MTT is the standard GLP-validated method, it indirectly measures viability via mitochondrial metabolism. Therefore, direct evaluation was performed using HCS analysis of nuclear morphology, which quantifies apoptotic cells at single-cell resolution through Hoechst staining and Boolean thresholding of nuclear area/size and fluorescence intensity. This detects condensed nuclei (~1,500 cells/well in controls), the final step of apoptosis before cell detachment. HCS quantification of viable cells identified the identical RA IC₅₀ of 119.9 µM (95% CI 101.2-149.3 µM; Fig. 1B), with representative images shown in Fig. 1C-E.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells