Mouse iPSC-derived Endothelial Cells
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
Mouse iPSC-derived endothelial cells (miPSC-ECs) are endothelial cells differentiated from mouse iPSCs. They are advantageous because they can be produced in large numbers and retain important characteristics of vascular endothelial cells. Inducing differentiation of mouse iPSCs, often through manipulation of BMP4, VEGF, and FGF2, results in cells that morphologically and phenotypically resemble endothelial cells.
Like most endothelial cells, miPSC-ECs are described to have a cobblestone phenotype when reaching confluence and express vascular endothelial markers such as CD31 (PECAM-1), VE-cadherin (CD144), and von Willebrand Factor (vWF). They have been shown to uptake acetylated low-density lipoprotein (Ac-LDL), form tube-like structures on Matrigel, and produce nitric oxide.
These cells offer an alternative to primary endothelial cell isolation which suffers from early senescence and high variability among donors. miPSC-ECs can be used to study vascular endothelial cell-specific functions including angiogenesis, barrier functions, and various cardiovascular disease mechanisms. When combined with iPSC lines generated from specific disease models, these cells can provide an alternative to animal heavy primary cell cultures to study disease mechanisms and screen for therapeutics.
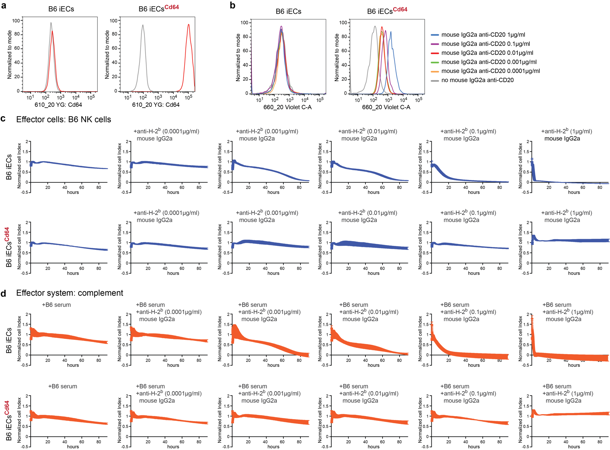
Cd64-expressing Mouse iECs are Protected from Antibody-mediated Killing
Allogeneic cell therapies often fail because the recipient's antibodies recognize and destroy the transplanted cells (via ADCC or CDC), significantly limiting their survival and efficacy. To develop a "cloaking" strategy by engineering cells to overexpress the IgG receptor CD64. This allows cells to capture host antibodies and block their killing mechanisms, enabling allogeneic grafts-including iECs, beta cells, and CAR T cells-to evade antibody-mediated destruction.
Mouse C57BL/6 (B6) induced pluripotent stem cells (iPSCs) were differentiated into B6 iECs, and the cells were transduced with lentiviral particles to express the mouse Cd64 transgene. These B6 iECsCd64 bound free IgG2a Fc in a dose-dependent manner (Fig. 1a, b). In impedance-based killing assays, Cd64 expression fully protected B6 iECs against ADCC and CDC mediated by anti-H-2b antibodies (Fig. 1c, d).
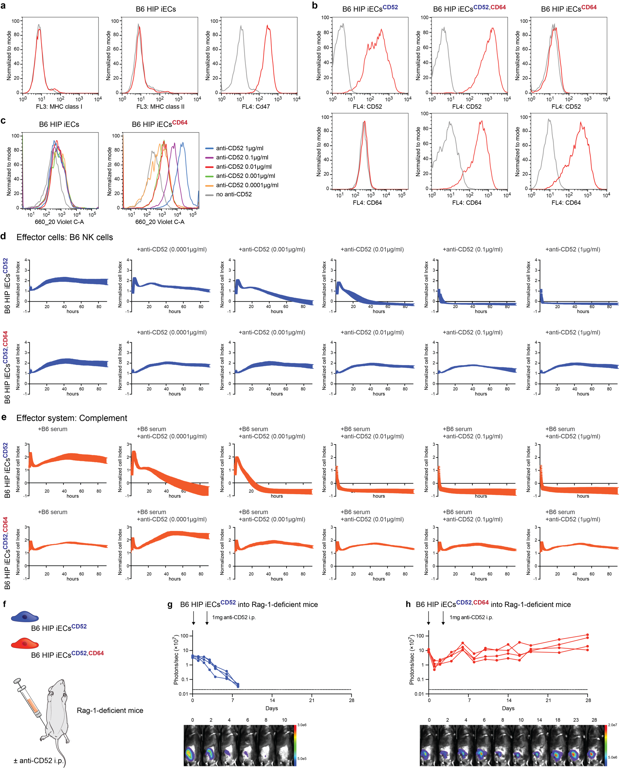
The strategy was then applied to B6 "hypoimmune" (HIP) iECs (*B2m-/-Ciita-/-*Cd47), which evade cellular immunity but remain vulnerable to antibodies (Fig. 2a). After adding human CD64 and the target antigen CD52 (Fig. 2b), these cells demonstrated robust human IgG1 Fc capture (Fig. 2c). While B6 HIP iECsCD52 were susceptible to alemtuzumab-mediated killing, B6 HIP iECsCD52,CD64 remained entirely resistant to both ADCC and CDC (Fig. 2d, e). In vivo, Luc+ grafts were transplanted into Rag1-/- mice (Fig. 2f). Following alemtuzumab treatment, B6 HIP iECsCD52 grafts were eliminated within one week, whereas B6 HIP iECsCD52,CD64 grafts survived, confirming that CD64 provides critical antibody protection in vivo (Fig. 2g, h).
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells