Immortalized Mouse Colonic Epithelial Cells-SV40
Cat.No.: CSC-I2180Z
Species: mouse
Morphology: Polygonal
Culture Properties: Adherent
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
free from contaminations (bacteria incl. mycoplasma, fungi, HIV, HAV, HBV, HCV, Parvo-B19) and cross-contaminations
Note: Never can cells be kept at -20°C.
The Immortalized Mouse Colonic Epithelial Cell Line represents a sophisticated in vitro model derived from primary murine colonic tissues, engineered via the stable integration of the Simian Virus 40 (SV40) Large T antigen. This strategic immortalization bypasses replicative senescence, ensuring a consistent and robust proliferative capacity without compromising the fundamental phenotypic characteristics of the colonic epithelium.
- Phenotypic Fidelity: These cells maintain the expression of critical epithelial markers and functional characteristics, including polarized morphology and the formation of tight junctions, making them an ideal surrogate for in vivo intestinal physiology.
- Extended Replicative Lifespan: Unlike primary cells that suffer from rapid "culture fatigue", the SV40-transformed line offers high-passage stability. This ensures experimental reproducibility and provides a cost-effective, continuous cell source for long-term studies.
- Versatile Research Applications: This line is a premier tool for investigating intestinal barrier function, electrolyte transport, mucosal immunology, and the pathogenesis of inflammatory bowel disease (IBD).
- Reliable Screening Platform: Given their consistent growth kinetics, these cells serve as a high-performance platform for drug toxicity assays and metabolic uptake studies, bridging the gap between basic research and preclinical development.
Our Immortalized Mouse Colonic Epithelial Cells undergo rigorous quality control, including testing for post-immortalization marker retention and sterility. By utilizing this model, researchers gain a high-fidelity, scalable system designed to accelerate discoveries in gastroenterology and epithelial biology.
Development and Characterization of Immortalized Mouse Intestinal Epithelial Cell Lines
Developing new tools for studying the intestinal epithelium aids in addressing unanswered questions in developmental biology, physiology, and disease pathology. Primary intestinal epithelial stem cell (ISC) cultures are an invaluable in vitro model system. However, their cultivation remains technically demanding and costly, limiting their accessibility and use. Here we derived immortalized intestinal epithelial cell lines (iIECs) from jejunal and colonic stem cell-enriched spheroid cultures from wild type C57BL/6 mice.
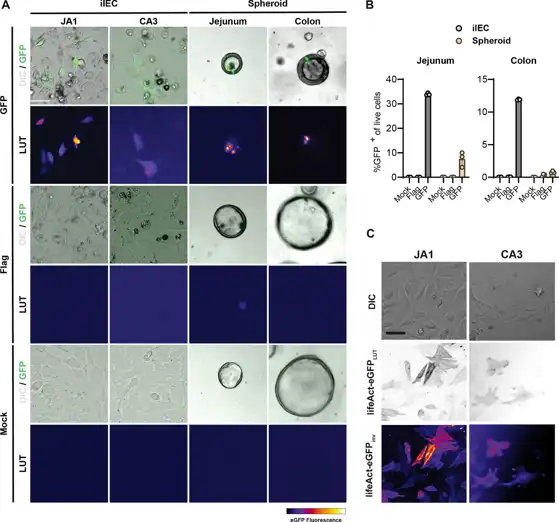
To evaluate the genetic tractability of iIECs, we conducted transient transfections using liposomal delivery of a control plasmid or a plasmid encoding a destabilized eGFP variant expressed under the control of a CMV promoter. Fluorescence microscopy performed 24 h post-transfection revealed heterogeneous cultures comprising a mix of GFP+ and GFP- cells (Fig. 1A). iIECs demonstrated significantly higher transfection efficiency than their primary 3D spheroid counterparts (Fig. 1B), indicating that iIECs can be productively utilized for transient transfection studies.
We next asked whether iIECs could be transduced using lentiviral vectors. Fluorescence imaging 48 h after transduction revealed robust labeling of both iIECJA1 (jejunal) and iIECCA3 (colonic) cell lines (Fig. 1C). These results demonstrate that iIEC lines are amenable to genetic manipulation using second-generation lentiviral vectors and are a genetically-tractable in vitro platform.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells
