Human iPSC-derived Neural Progenitor Cells
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
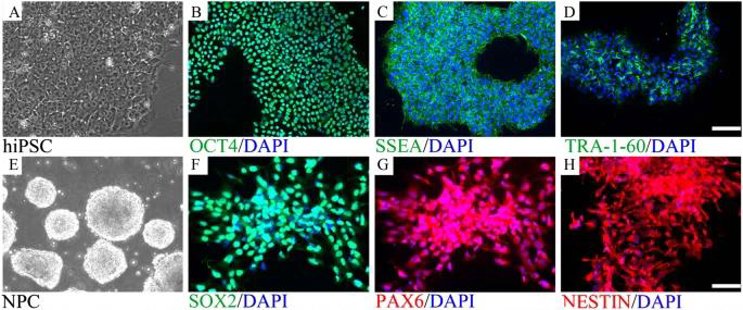
Human iPSC-derived Neural Progenitor Cells (hiPSC-NPCs) are a specialized cell lineage generated through the directed differentiation of human induced Pluripotent Stem Cells (hiPSCs), which are reprogrammed from somatic cells (e.g., fibroblasts, peripheral blood mononuclear cells) via the introduction of pluripotency-associated transcription factors (e.g., OCT4, SOX2, KLF4, c-MYC). Functionally, hiPSC-NPCs possess two key properties: self-renewal (proliferating long-term without losing differentiation potential) and multilineage differentiation capacity (giving rise to all major neural cell types, including neurons, astrocytes, and oligodendrocytes).
These properties make hiPSC-NPCs a versatile tool for a variety of applications: In neurodevelopment, they can be used as in vitro model to study human neural development, which for the first time gives experimental access to processes which cannot be studied in vivo (e.g. lineage specification, maturation, etc.). In disease modeling of neurodegenerative diseases, patient-specific hiPSC-NPCs can be used to recapitulate disease specific phenotypes (abnormal protein aggregation in Alzheimer's disease, loss of motor neurons in ALS, etc.) and can then be used to study the pathogenic mechanisms in detail and identify novel drug targets. In drug discovery, hiPSC-NPCs can be used as a human relevant model for high-throughput drug screening of neuroprotective compounds. Finally, in regenerative medicine, they have been shown in preclinical studies to be a potential candidate for transplantation-based therapies where they may be used to replace damaged neural cells in diseases such as spinal cord injury or stroke to aid in tissue repair and functional recovery.
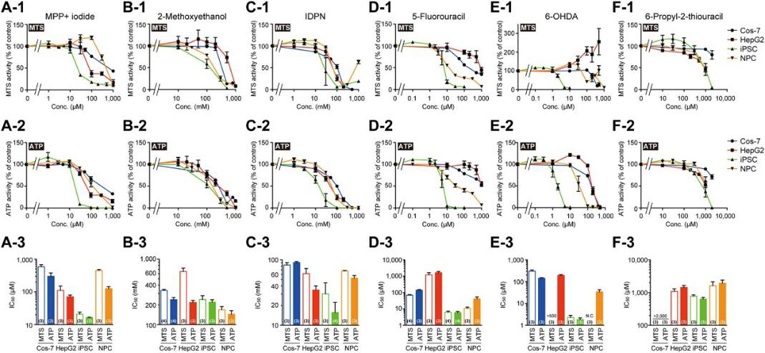
Cytotoxicity of 35 DNT chemicals on Cos-7, HepG2, iPSC, and NPC
The OECD test guideline 426 for assessing developmental neurotoxicity (DNT) of chemicals relies heavily on animal testing, which is costly, time-consuming, and raises ethical concerns. Kamata et al. presents an alternative protocol using human-induced pluripotent stem cells (iPSC) and their differentiation into neural progenitor cells (NPC) to evaluate DNT in a simpler, quicker, and less expensive manner.
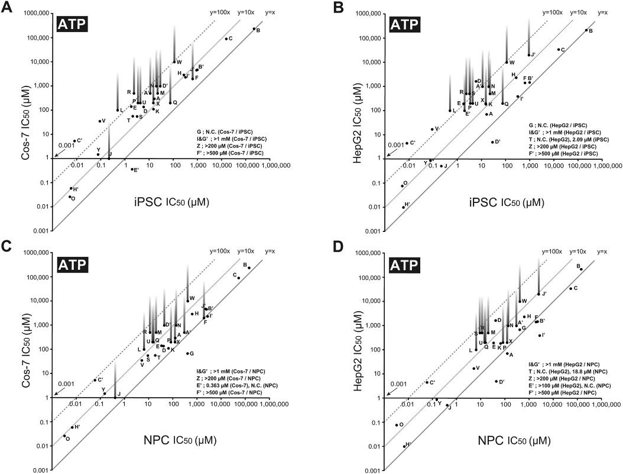
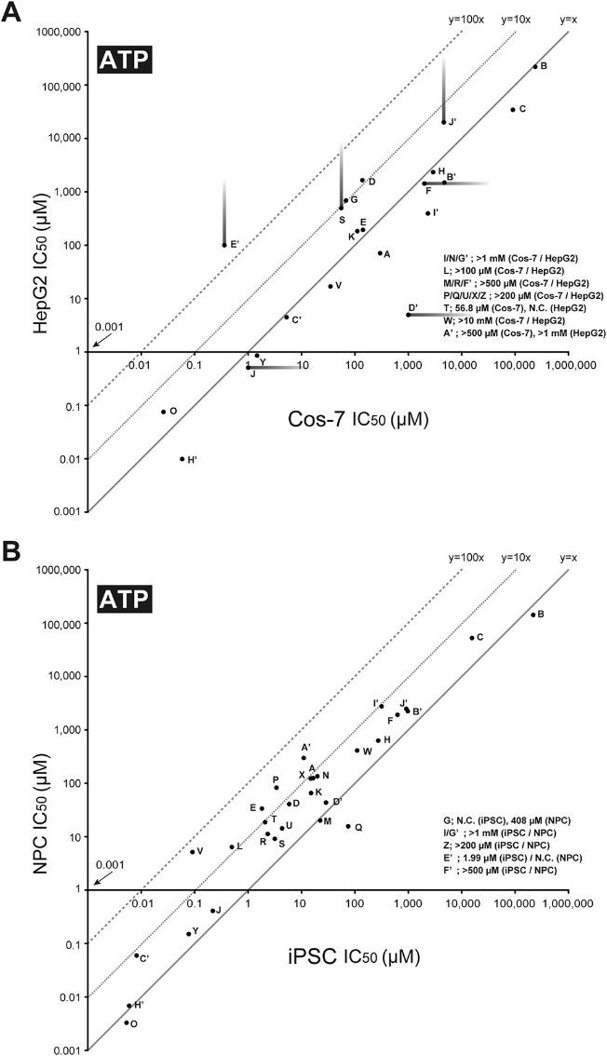
Thirty-five DNT chemicals and acetaminophen (negative control) were tested on four cell types (Cos-7, HepG2, iPSC, and NPC) at various concentrations. After two days, cell survival was assessed using mitochondrial MTS reduction and cellular ATP level assays, and IC50 values were calculated (Fig. 1). Most DNT chemicals showed concentration-dependent inhibition, though some, like aldicarb, n-hexane, thalidomide, and toluene, were not cytotoxic at their highest concentrations. The IC50 values for most DNT chemicals were significantly lower in iPSC and NPC compared to Cos-7 and HepG2 cells (Fig. 2). Differences in IC50 values were more pronounced between Cos-7 and HepG2 cells than between iPSC and NPC (Fig. 3). NPC passage numbers did not affect the results.
NPC Surface Modification with Esbp-PEG-lipid
Cell-based therapy offers a potential solution for stroke-related disorders, which have limited treatment options after 4.5 hours. Goel et al. explored enhancing stroke treatment using NPCs modified with Esbp-PEG-lipid. NPCs were surface-modified with Esbp-PEG-lipid of different PEG molecular weights (5 and 40 kDa) and optimized for binding to stroke area surfaces, such as recombinant E-selectin and TNF-α activated endothelium.
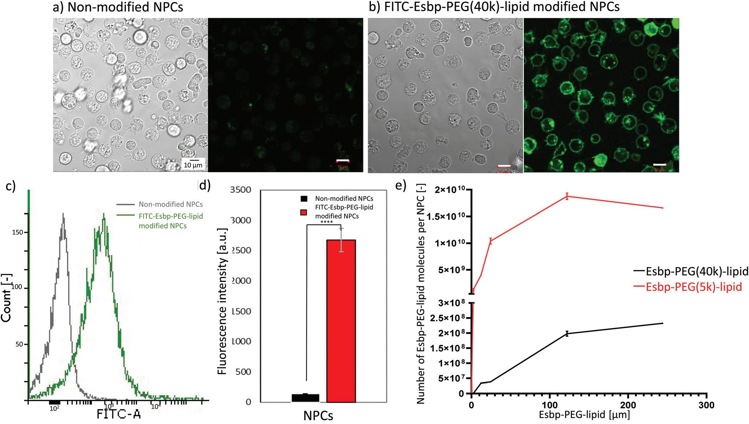
To confirm surface modification, FITC-Esbp-PEG-lipid-modified cells were analyzed by flow cytometry and confocal microscopy, and the number of Esbp molecules on the cell surface was quantified. Confocal images (Fig. 4(a) and (b)) showed green fluorescence from FITC on modified NPCs, but not on unmodified cells. Quantitative measurements (Fig. 4(c) and (d)) revealed a 50-fold increase in fluorescence intensity in treated NPCs compared to untreated cells, indicating successful incorporation of Esbp onto the NPC surface via PEG-lipid. The number of incorporated FITC-Esbp-PEG-lipids (Fig. 4(e)) depended on the feed concentration, with a maximum of approximately 2.0 × 108 molecules for 40k and 2.0 × 1010 for 5k at concentrations above 120 µM. At 25 µM, the number of incorporated molecules was about 4.0 × 107 for 40k and 1.0 × 1010 for 5k. These numbers were deemed sufficient for surface modification, so these cells were used for further experiments. The significant difference in the number of incorporated 5k and 40k PEG lipid molecules may be due to the larger size of 40k PEG causing more steric hindrance on cell surfaces.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells