HCA-7 Colony 29
Cat.No.: CSC-C9447J
Species: Homo sapiens (Human)
Source: Intestine; Colon
- Specification
- Background
- Scientific Data
- Q & A
- Customer Review
HCA-7 Colony 29 (HCA-7 Col.29) is an established human colorectal adenocarcinoma cell line derived from a primary colon carcinoma. It is a model system that displays a well-differentiated epithelial tumor phenotype and is used as an in vitro model to study colorectal cancer biology, including signaling pathways associated with inflammation-driven tumorigenesis.
HCA-7 Colony 29 cells are adherent monolayer culture cells with cobblestone-like epithelial morphology and exhibit tight intercellular junctions. The hallmark feature of this cell line is the high constitutive expression of cyclooxygenase-2 (COX-2) and the robust production of prostaglandin E₂ (PGE₂), which mimics the inflammatory signaling observed in colorectal tumors. Additionally, these cells express epithelial markers such as cytokeratins and E-cadherin, indicating a retained epithelial differentiation. HCA-7 Colony 29 is used extensively to study the role of COX-2-prostaglandin signaling in colorectal cancer progression, tumor-associated inflammation, angiogenesis, and immune modulation. It is also a well-established model for the screening of nonsteroidal anti-inflammatory drugs (NSAIDs), selective COX-2 inhibitors, and other anti-cancer or anti-inflammatory agents.
P-Cadherin Knockout Accelerates IEC Wound Healing by Modulating Cell-Matrix Adhesion and Cell Spreading
Inflammatory bowel diseases (IBD) cause recurrent intestinal inflammation, disrupting epithelial homeostasis and potentially leading to colitis-associated colon cancer (CAC). P-cadherin, an adhesion protein upregulated in inflamed mucosa, may play a role in intestinal inflammation and CAC. Naydenov et al. investigated P-cadherin's roles in these processes.
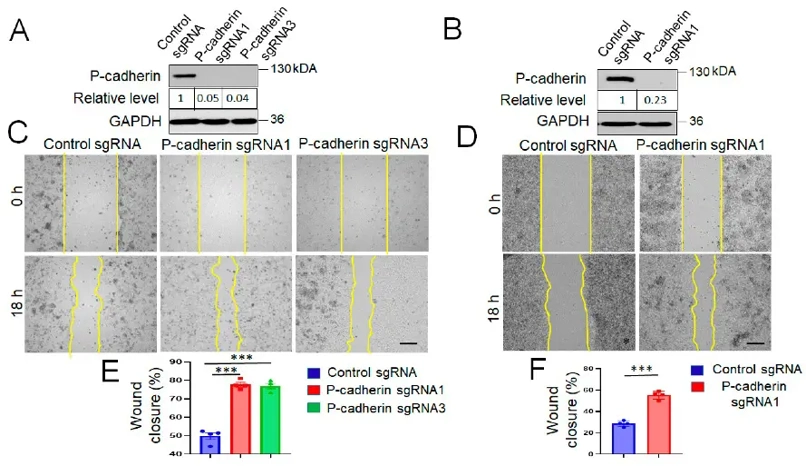
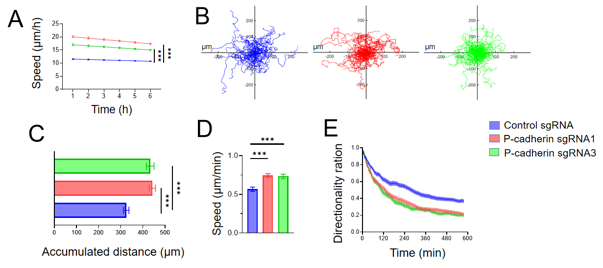
They investigated the role of P-cadherin in modulating intestinal epithelial cell (IEC) migration using HCA-7 Colony 29 (referred hereafter as HCA-7) - and SK-CO15 cells, well-differentiated human colonic carcinoma cell lines. CRISPR-Cas9 was used to knockout P-cadherin in these cell lines. The loss of P-cadherin did not significantly affect cell proliferation but significantly enhanced collective IEC migration in wound healing models (Fig. 1C-F). Live imaging of wounded HCA-7 monolayers showed higher migration velocity in P-cadherin knockout cells compared to controls (Fig. 2A). Individual cell migration analysis revealed increased migrated distance and speed but decreased directionality in P-cadherin-deficient cells (Fig. 2B-E). These findings suggest that P-cadherin directly affects cell motility, crucial for wound healing, rather than indirectly influencing it through junctional remodeling.
.
Ask a Question
Write your own review
- Adipose Tissue-Derived Stem Cells
- Human Neurons
- Mouse Probe
- Whole Chromosome Painting Probes
- Hepatic Cells
- Renal Cells
- In Vitro ADME Kits
- Tissue Microarray
- Tissue Blocks
- Tissue Sections
- FFPE Cell Pellet
- Probe
- Centromere Probes
- Telomere Probes
- Satellite Enumeration Probes
- Subtelomere Specific Probes
- Bacterial Probes
- ISH/FISH Probes
- Exosome Isolation Kit
- Human Adult Stem Cells
- Mouse Stem Cells
- iPSCs
- Mouse Embryonic Stem Cells
- iPSC Differentiation Kits
- Mesenchymal Stem Cells
- Immortalized Human Cells
- Immortalized Murine Cells
- Cell Immortalization Kit
- Adipose Cells
- Cardiac Cells
- Dermal Cells
- Epidermal Cells
- Peripheral Blood Mononuclear Cells
- Umbilical Cord Cells
- Monkey Primary Cells
- Mouse Primary Cells
- Breast Tumor Cells
- Colorectal Tumor Cells
- Esophageal Tumor Cells
- Lung Tumor Cells
- Leukemia/Lymphoma/Myeloma Cells
- Ovarian Tumor Cells
- Pancreatic Tumor Cells
- Mouse Tumor Cells