In Vitro Permeability and Transporters
Creative Bioarray provides a number of in vitro permeability and transporter assays to evaluate drug permeability and predict drug absorption and distribution, including Caco-2 permeability assay, MDCK permeability assay, Parallel artificial membrane permeability assay (PAMPA), and a panel of transporter assays on several of the most relevant transporters such as canine P-glycoprotein (P-gp), BCRP, BSEP, OAT1, OAT3, OATP1B1, OATP1B3, OCT1, and OCT2.
Prediction of drug absorption is an important goal in the design, optimization, and selection of drugs. Drug permeability across biological membranes is a key factor in the absorption of drugs across gastrointestinal mucosa and the distribution of drugs to tissues through blood circulation. Poor permeability can lead to poor absorption (for orally administered drugs) or poor distribution throughout the body. The permeability of a drug across a membrane depends on both the passive diffusion and the active transport of the drug.
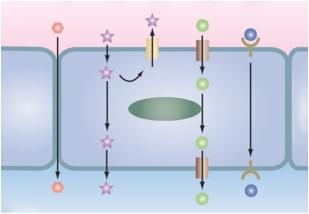
There are a number of in vitro cell-based or cell-free methods to study drug permeability and transporter interactions:
Caco-2 permeability assay is the gold standard method to evaluate both the passive and active transport and the absorption of orally taken drugs. The method can be used to assess the bidirectional permeability of a test compound (from the apical to the basolateral side or vice versa).
MDCK permeability assay is a useful tool for the identification of substrates and inhibitors of efflux transporters P-gp, and for the calculation of net flux ratio.
PAMPA, a cell-free method that uses a lipid-infused artificial membrane to determine the passive permeability of substances from a donor compartment to an acceptor compartment, is a widely used assay to predict oral absorption that exhibit a high degree of correlation with Caco-2 assay. Mechanism of drug absorption can be better estimated combining the results of PAMPA and of Caco-2 assay.
Other related assays and transporter assays are also available. We provide assays on a variety of transporters, including but not limited to P-gp, BCRP, BSEP, OAT1, OAT3, OATP1B1, OATP1B3, OCT1, and OCT2. Contact us for more information.
Creative Bioarray is staffed with experienced experts in the field of drug development. We are dedicated to serving our customers with well-developed assays and high quality data, helping our customers achieve a trouble-free drug screening and optimizing experience. We are capable of tailoring the assays in order to meet your needs. If you have any questions, please feel free to contact us. We look forward to working with you in the future.
Explore Other Options
For research use only. Not for any other purpose.
Services
-
Cell Services
- Cell Line Authentication
- Cell Surface Marker Validation Service
-
Cell Line Testing and Assays
- Drug-Resistant Cell Models
- Cell Viability Assays
- Cell Proliferation Assays
- Cell Migration Assays
- Soft Agar Colony Formation Assay Service
- SRB Assay
- Cell Apoptosis Assays
- Cell Cycle Assays
- Cell Angiogenesis Assays
- DNA/RNA Extraction
- Custom Cell & Tissue Lysate Service
- Cellular Phosphorylation Assays
- Stability Testing
- Sterility Testing
- Endotoxin Detection and Removal
- Phagocytosis Assays
- Cell-Based Screening and Profiling Services
- 3D-Based Services
- Custom Cell Services
- Cell-based LNP Evaluation
-
Stem Cell Research
- iPSC Generation
- iPSC Characterization
-
iPSC Differentiation
- Neural Stem Cells Differentiation Service from iPSC
- Astrocyte Differentiation Service from iPSC
- Retinal Pigment Epithelium (RPE) Differentiation Service from iPSC
- Cardiomyocyte Differentiation Service from iPSC
- T Cell, NK Cell Differentiation Service from iPSC
- Hepatocyte Differentiation Service from iPSC
- Beta Cell Differentiation Service from iPSC
- Brain Organoid Differentiation Service from iPSC
- Cardiac Organoid Differentiation Service from iPSC
- Kidney Organoid Differentiation Service from iPSC
- GABAnergic Neuron Differentiation Service from iPSC
- Undifferentiated iPSC Detection
- iPSC Gene Editing
- iPSC Expanding Service
- MSC Services
- Stem Cell Assay Development and Screening
- Cell Immortalization
-
ISH/FISH Services
- In Situ Hybridization (ISH) and RNAscope Services
- Fluorescent In Situ Hybridization
- FISH Probe Design, Synthesis and Testing Service
-
FISH Applications
- Multicolor FISH (M-FISH) Analysis
- Chromosome Analysis of ES and iPS Cells
- RNA FISH in Plant Service
- Mouse Model and PDX Analysis (FISH)
- Cell Transplantation Analysis (FISH)
- In Situ Detection of CAR-T Cells & Oncolytic Viruses
- CAR-T/CAR-NK Target Assessment Service (ISH)
- ImmunoFISH Analysis (FISH+IHC)
- Splice Variant Analysis (FISH)
- Telomere Length Analysis (Q-FISH)
- Telomere Length Analysis (qPCR assay)
- FISH Analysis of Microorganisms
- Neoplasms FISH Analysis
- CARD-FISH for Environmental Microorganisms (FISH)
- FISH Quality Control Services
- QuantiGene Plex Assay
- Circulating Tumor Cell (CTC) FISH
- mtRNA Analysis (FISH)
- In Situ Detection of Chemokines/Cytokines
- In Situ Detection of Virus
- Transgene Mapping (FISH)
- Transgene Mapping (Locus Amplification & Sequencing)
- Stable Cell Line Genetic Stability Testing
- Genetic Stability Testing (Locus Amplification & Sequencing + ddPCR)
- Clonality Analysis Service (FISH)
- Karyotyping (G-banded) Service
- Animal Chromosome Analysis (G-banded) Service
- I-FISH Service
- Molecular Karyotyping (aCGH)
- Droplet Digital PCR (ddPCR) Service
- Digital ISH Image Quantification and Statistical Analysis
- SCE (Sister Chromatid Exchange) Analysis
- Biosample Services
- Histology Services
- Exosome Research Services
- Drug Metabolism and Pharmacokinetics (DMPK)
-
Safety Evaluation Services
- High-Throughput Toxicity Screening
- High-Content Cytotoxicity Screening
- In Vitro Cardiotoxicity
- In Vitro Genotoxicity
- In Vitro Hepatotoxicity
- In Vitro Neurotoxicity
- In Vitro Nephrotoxicity
- In Vitro Dermal Toxicology
- Ocular Toxicity
- In Vitro Cytotoxicity
- Endocrine Disruption Screening Assay
- In Vivo Toxicity Study