In Vitro Mammalian Chromosomal Aberration Test (OECD 473)
Creative Bioarray provides in vitro mammalian chromosomal aberration test (OECD 473) to identify substances that cause structural chromosomal aberrations in cultured mammalian cells (Chinese Hamster Ovary cell line CHO-K1, other cells upon request). This assay is available under either GLP (Good Laboratory Practice) or Non-GLP conditions.
Many human genetic diseases are caused by chromosomal aberrations. Chromosomal damages in somatic cells that involve alterations in oncogenes and tumor suppressor genes are also related to tumor formation in humans and animals. Therefore, in vitro mammalian chromosomal aberration test is one of the recommended in vitro tests for mutagen assessment.
This test is not designed to measure aneuploidy. Although aneuploidy may arise in the chromosomal aberration assay, aneuploidy alone doesn’t indicate that the test article has aneugenic potential. For the detection of aneuploidy, we recommend that you choose in vitro mammalian cell micronucleus test (OECD 487).
Test Method:
The in vitro mammalian chromosomal aberration test is carried out according to the OECD Guideline 4731. Proliferating mammalian cell cultures (e.g. Chinese Hamster Ovary cell line CHO or CHO-K1, Human Peripheral Blood Lymphocytes HPBL) are exposed to the test chemical both with and without an exogenous source of metabolic activation (Aroclor-1254 induced rat liver S9). After an appropriate period of exposure (short term exposure or long term exposure) to the test articles (at least 3 concentrations with duplicates), the cells are treated with a metaphase-arresting substance (e.g. Colcemid or colchicine), and harvested. Chromosomal spread slides are then prepared and stained. Metaphase cells are analyzed for the presence of chromosomal aberrations under microscopy. Negative vehicle control and positive controls are included in each study.
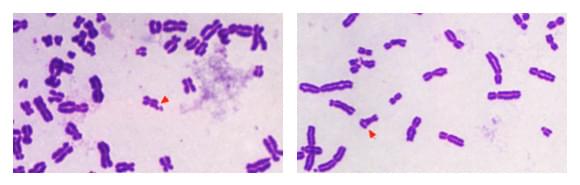
Figure 1. Chromosomal aberrations
Creative Bioarray can provide in vitro mammalian chromosomal aberration test following GLP principles2. Typically, a draft study plan is prepared and reviewed by the sponsor and our quality assurance (QA) personnel after the collection of the characterization and formulation information of the test articles. The study can get started after the approval of the study plan by both parties.
For test compounds with no toxicity or solubility information, we will perform range-finding tests before the main assay (additional fees may be charged).
After the study, a draft report of the results will be prepared for the sponsor and the quality assurance group to review. After the final approval, the final report will be provided with a QA statement.
To find out more about our service with more information, please feel free to leave a message below, or contact us. Our well-experienced experts will get back to you with required information.
References:
- OECD Guideline for Testing of Chemicals: In Vitro Mammalian Chromosomal Aberration Test. TG 473. Adopted 29 July 2016.
- OECD Principles of Good Laboratory Practice (as revised in 1997). OECD Environmental Health and Safety Publications. OECD. 1. 1998.

