In Vitro Mammalian Cell Micronucleus Test (OECD 487)
Creative Bioarray provides in vitro mammalian cell micronucleus test (OECD 487), a genotoxicity test to detect microneuclei in the cytoplasm of interphase cells and to identify genotoxic substances that harbor chromosome damaging potential in vitro. Aneugens and clastogens can be detected by analyzing the frequency of microneuclei in cells that have undergone cell mitosis during or after exposure to the test chemical. This assay is available under either GLP (Good Laboratory Practice) or Non-GLP conditions.
Micronuclei are small nuclei that separates from the main nuclei of cells, produced during telophase of mitosis or meiosis by lagging chromosome fragments or whole chromosomes, and are not incorporated into one of the daughter nuclei. Therefore, the presence of micronuclei can serve as a sign of genotoxicity and chromosomal instability. The in vitro mammalian cell micronucleus test is designed to assess the chromosome damaging potential of test articles.
This test can be carried out in a variety of cell types, including cell lines and primary cell cultures of human or rodent origin.
Test System:
- Chinese Hamster Ovary cell line CHO-K1
- Chinese Hamster Lung cell line V79
- TK6 Human Lymphoblastoid cells
- Primary Human Lymphocytes
Test Method:
The in vitro mammalian cell micronucleus test is carried out in compliance with the OECD Guideline 4871. Proliferating mammalian cell cultures (e.g. CHO-K1, or Human Lymphocytes) are exposed to the test chemical for an indicated length of time (3-6 hours or throughout the test) both in the presence and in the absence of an exogenous source of metabolic activation (Aroclor-1254 induced rat liver S9). After an appropriate period of exposure (short term exposure or long term exposure) to the test articles (at least 3 concentrations with duplicates/triplicates), the cells are grown for sufficient time (1.5-2.0 normal cell cycle lengths from the beginning of treatment) to allow chromosome damage to take place and to form micronuclei in the cells. Cells are harvested for analyzing by flow cytometry or by microscope. For microscopy examination, chromosomal spread slides are prepared, stained, and analyzed for the presence and number of micronuclei. Negative vehicle control and positive controls are included in each study.
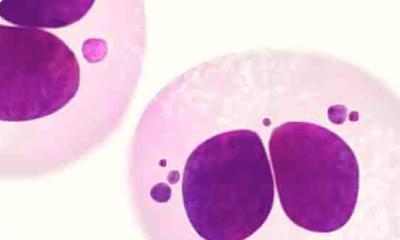
Figure 1. Presence of micronuclei next to the daughter nuclei
Creative Bioarray can provide in vitro mammalian cell micronucleus test following GLP principles2. Typically, a draft study plan is prepared and reviewed by the sponsor and our quality assurance (QA) personnel after the collection of the characterization and formulation information of the test articles. The study can get started after the approval of the study plan by both parties.
For test compounds with no toxicity or solubility information, we will perform range-finding tests before the main assay (additional fees may be charged).
After the study, a draft report of the results will be prepared for the sponsor and the quality assurance group to review. After the final approval, the final report will be provided with a QA statement.
To find out more about our service with more information, please feel free to leave a message below, or contact us. Our well-experienced experts will be more than happy to help.
References:
- OECD Guideline for Testing of Chemicals: In Vitro Mammalian Cell Micronucleus Test. TG 487. Adopted 26 September 2014.
- 2. OECD Principles of Good Laboratory Practice (as revised in 1997). OECD Environmental Health and Safety Publications. OECD. 1. 1998.

