In Vitro Metabolism
With extensive knowledge and experience in drug discovery, Creative Bioarray can provide standard, cost-effective in vitro metabolism services, including drug metabolic stability services and drug-drug interaction services to support your drug development process.
Drug metabolism affects drug safety and efficacy
Drug safety and efficacy, the essential issues in drug discovery, are greatly affected by drug metabolism and drug-drug interactions. Since a drug is eliminated either by excretion or by metabolism to one or more active or inactive metabolites after entering the body, the metabolism rate and the bioactivity of the metabolites generated in the elimination process directly affect the retaining time of the drug in the body in bioactive forms. A high metabolism rate and the generation of active metabolites can produce a blood level that causes unacceptable toxicity. Therefore, it is crucial to understand better the metabolizing route of a drug at an early stage of drug development to predict drug toxicity or side effects, drug-drug interactions and obtain information that helps form dosing strategies.
The primary goals of in vitro drug metabolism evaluation are:
- To identify major metabolic pathways and enzymes that affect the elimination of the test drug and its metabolites;
- To evaluates the effects of the test drug on the metabolism of other drugs, or vice versa
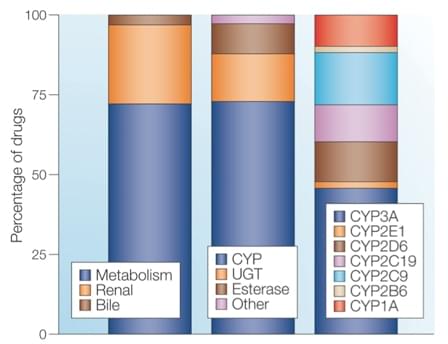 Figure 1. Routes of elimination of the top 200 most prescribed drugs in 2002. Metabolism represents the listed clearance mechanism for ~73% of the top 200 drugs. About three-quarters of the drugs cleared via metabolism are metabolized by members of the cytochrome P450 (CYP) superfamily.
Figure 1. Routes of elimination of the top 200 most prescribed drugs in 2002. Metabolism represents the listed clearance mechanism for ~73% of the top 200 drugs. About three-quarters of the drugs cleared via metabolism are metabolized by members of the cytochrome P450 (CYP) superfamily.
Creative Bioarray provides various in vitro metabolism assays
- Metabolite Profiling and Identification
- Metabolic Stability Assays
- Hepatocyte stability assay
- S9 stability assay
- Microsomal stability assay
- Plasma stability assay
- Recombinant enzymes stability assay
- Drug-Drug Interaction Assays (DDI)
Due to the complexity of the drug development process, you need a flexible partner to provide reliable support to your specific needs. With extensive experience and knowledge in drug development, Creative Bioarray can provide various flexible and integrated services for drug discovery. Our experienced scientific team and state-of-the-art facilities enable us to offer our clients the following benefits:
- Flexibility to customize assays
- Reliable data of high consistency and quality
- Highly competitive pricing
- Detailed, concise report of results
Creative Bioarray is dedicated to providing fast and integrated services of the highest quality to accelerate drug discovery. If you have any special needs or questions regarding our services, please feel free to contact us to get support from our experienced experts. We look forward to working with you in the future.
References
- US Food and Drug Administration. "Guidance for industry: Drug metabolism/drug interaction studies in the drug development process: studies in vitro." US Food and Drug Administration, Rockville, MD (1997).
- Wienkers, Larry C., and Timothy G. Heath. "Predicting in vivo drug interactions from in vitro drug discovery data." Nature reviews Drug discovery 4.10 (2005): 825-833.

